|
Recherches sur la spectroscopie par transformations de Fourier, Éd. Monthly Notices of the Royal Astronomical Society, 118, 224–233.Ĭonnes, J. Equivalent quantum-efficiencies of photographic emulsions. The luminosity of spectrometers with prisms, gratings, or Fabry-Perot etalons. Philosophical Transactions of the Royal Society of London, 90, 255–283. To which is added, an inquiry into the method of viewing the sun advantageously, with telescopes of large apertures and high magnifying powers. Investigation of the powers of the prismatic colours to heat and illuminate objects with remarks, that prove the different refrangibility of radiant heat. Red-green emitting and superparamagnetic nanomarkers containing Fe3O4 functionalized with calixarene and rare earth complexes. Physical Chemistry Chemical Physics, 19, 18660–18670. Efficient multicolor tunability of ultrasmall ternary-doped LaF 3 nanoparticles: Energy conversion and magnetic behavior. It is always a good idea to compare your IR spectrum with an authentic spectrum of the material you think you have.Shrivastava, N., Khan, L., Vargas, J., Ospina, C., Coaquira, J., Zoppellaro, G., Brito, H., Javed, Y., Shukla, D., & Felinto, M. The operating system often has the capability of searching one or more databases of spectra and finding the spectra that most closely match the spectrum that was just run. 
All modern FT-IR spectrometers are controlled by computers. They are also available in computer readable format for rapid searching and spectrum matching. Two of the most popular collections are the Sadtler Index of IR Spectra and the Aldrich Library of Infra-red Spectra Both collections are easily accessible in 'hard copy' form in most major university libraries. Reference Spectra The Infrared spectra of thousands of compounds have been determined and compiled by several different companies. This technique is called running a "neat" spectrum, meaning the spectrum is of the pure liquid only, without solvent. Excellent spectra can be obtained in a matter of a few minutes with minimum expense. A pure sample of the liquid (1-2 drops) may be placed between two disks of pure NaCl or KBr and the resulting 'sandwich' placed directly in the sample holder of the spectrometer. The resulting 'KBr Disk' will produce a spectrum free of almost all extraneous peaks. Solid spectra may also be obtained by mixing the solid with dry KBr, grinding to a fine, well mixed powder, and then forming a disk of the mixture by applying high pressure in a specially designed device. The NUJOL exhibits only a very few well defined peaks that can be ignored when examining the spectrum of the mull. A solid could also be ground to a fine paste with NUJOL TM (a mixture of highly purified hydrocarbons) and the resulting 'mull' studied directly. IR spectra of solids and liquids are usually obtained by dissolving the sample in a relatively IR transparent solvent such as CCl 4 and using simple liquid cells. The only draw-back is that it is very expensive and delicate cells are needed. IR gas analysis is a common analytical tool for those involved in studies of atmospheric pollution. Be sure to read that section of your text before doing your first IR so that you will understand what you are doing.įigure 2 : Close-up of the salt plates in the IR instrument, ready to take a sample Sampling IR spectra can be determined for solids, liquids, or gases. Every modern Organic text discusses IR theory in some detail. To save space, we will not discuss the theory of IR Spectroscopy. In fact, the IR of a pure compound bears the same relationship to that compound as fingerprints do to an individual. It is commonly used in judicial proceedings as much as fingerprints are used. Evidence provided by IR is widely respected. The technique is simple and can often provide a definitive answer in less than ten minutes. The quickest and easiest way to determine the presence of one of these "Functional Groups" is to take the IR spectrum of the compound. 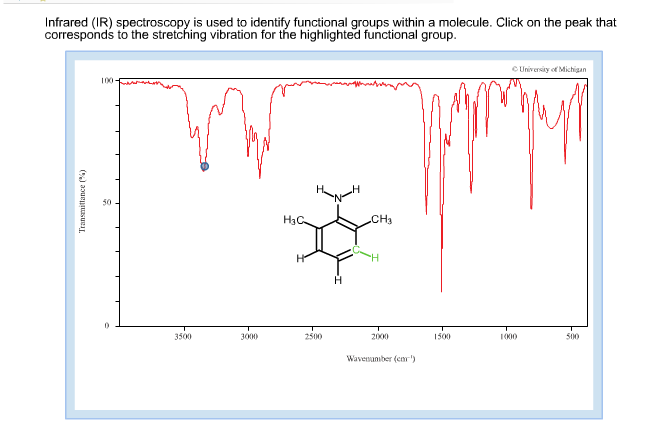
As you will learn, IR may actually be thought of as a Functional Group detector. Alkenes contain a carbon-carbon double bond (C=C), and ketones contain a carbon-oxygen double bond (C=O). For example, all alcohols contain an 'O-H' group attached to an sp 3 hybridized carbon atom. Each of these classes is distinguished by the presence of a "Functional Group" in the molecule. 
Introduction During the course of this year we will study several different classes of compounds including alcohols, alkenes, and ketones. 4 To identify a compound by an investigation of its infrared spectrum
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
